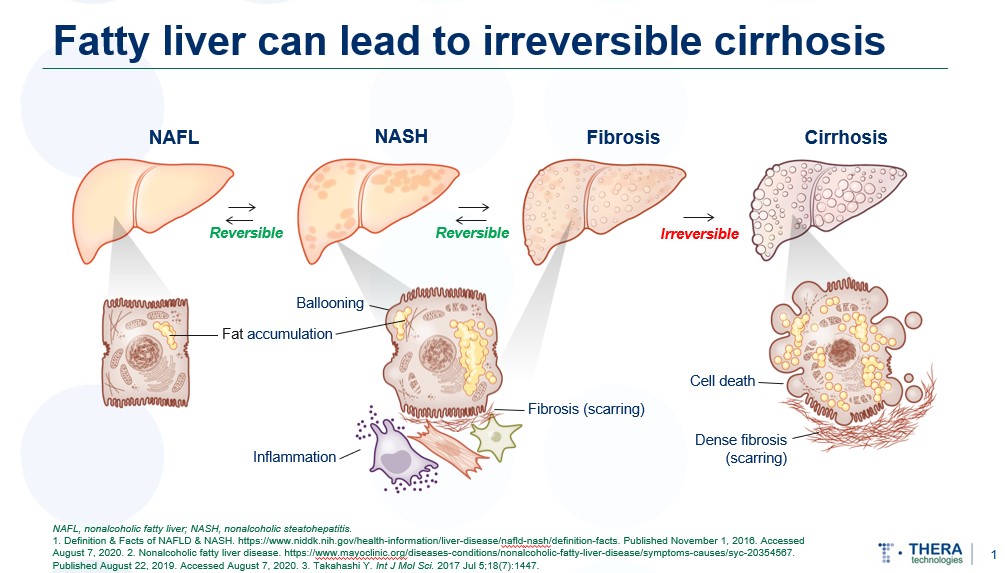
In a Phase 2 clinical study in people with HIV (PWH) with NAFLD, tesamorelin significantly decreased liver steatosis by 37%9, 14. A high percentage of subjects (60%) were able to achieve 30% decrease of liver fat reduction after 1 year of treatment compared to placebo subjects (16%, internal data). Tesamorelin also demonstrated an impact on liver fibrosis by significantly reducing fibrosis progression compared to placebo group14. In addition, the results of the study demonstrated critical changes associated with tesamorelin treatment relative to placebo in key liver gene sets, including reduction in hepatic fat oxidation, and reduction in key inflammatory and fibrotic pathways13.
- Anstee, Q.M., et al., From NASH to HCC: current concepts and future challenges. Nat Rev Gastroenterol Hepatol, 2019.
- Lim, S., M.R. Taskinen, and J. Boren, Crosstalk between nonalcoholic fatty liver disease and cardiometabolic syndrome. Obes Rev, 2018.
- Estes, C., et al., Modeling NAFLD disease burden in China, France, Germany, Italy, Japan, Spain, United Kingdom, and United States for the period 2016-2030. J Hepatol, 2018. 69(4): p. 896-904.
- Swain, M.G., et al., Burden of nonalcoholic fatty liver disease in Canada, 2019-2030: a modelling study. CMAJ Open, 2020. 8(2): p. E429-E436.
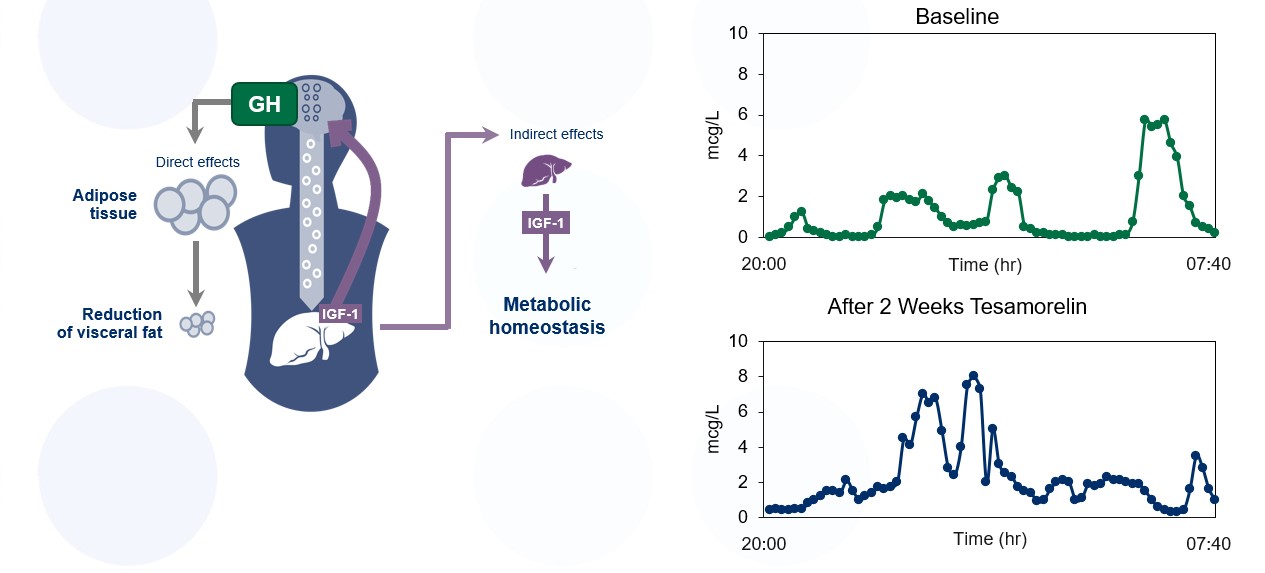
- Stanley, T.L., et al., Effects of a growth hormone-releasing hormone analog on endogenous GH pulsatility and insulin sensitivity in healthy men. J Clin Endocrinol Metab, 2011. 96(1): p. 150-158.
- Serono, E., Serostim® Product Monograph. 2007.
- Casanueva, F.F., Physiology of growth hormone secretion and action. Endocrinol. Metab Clin. North Am, 1992. 21(3): p. 483-517.
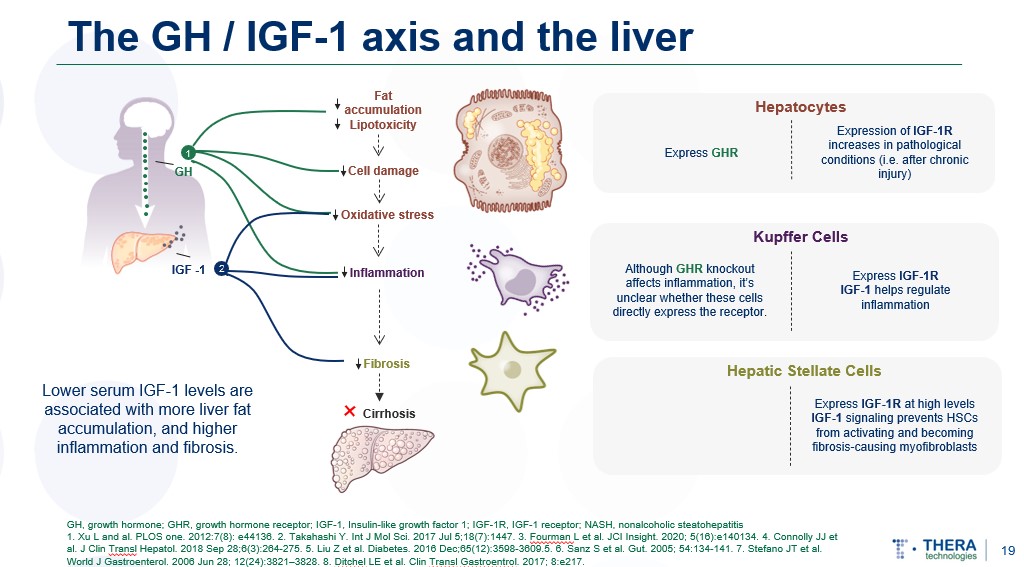
- Liu, Z., et al., Growth Hormone Control of Hepatic Lipid Metabolism. Diabetes, 2016. 65(12): p. 3598-3609.
- Stanley, T.L., et al., Effect of Tesamorelin on Visceral Fat and Liver Fat in HIV-Infected Patients With Abdominal Fat Accumulation: A Randomized Clinical Trial. Jama, 2014. 312(4): p. 380-389.
- Xu, L., et al., Association between serum growth hormone levels and nonalcoholic fatty liver disease: a cross-sectional study. PLoS One, 2012. 7(8): p. e44136.
- Dichtel, L.E., et al., The Association Between IGF-1 Levels and the Histologic Severity of Nonalcoholic Fatty Liver Disease. Clin Transl Gastroenterol, 2017. 8(1): p. e217.
- Takahashi, Y., The Role of Growth Hormone and Insulin-Like Growth Factor-I in the Liver. Int J Mol Sci, 2017. 18(7).
- Fourman, L.T., et al., Effects of tesamorelin on hepatic transcriptomic signatures in HIV-associated NAFLD. JCI Insight, 2020. 5(16).
- Stanley, T.L., et al., Effects of tesamorelin on non-alcoholic fatty liver disease in HIV: a randomised, double-blind, multicentre trial. The Lancet HIV, 2019.
1 Definition & Facts of NAFLD & NASH. https://www.niddk.nih.gov/health-information/liver-disease/nafld-nash/definition-facts. Published November 1, 2016. Accessed August 7, 2020.
2 Nonalcoholic fatty liver disease. https://www.mayoclinic.org/diseases-conditions/nonalcoholic-fatty-liver-disease/symptoms-causes/syc-20354567. Published August 22, 2019. Accessed August 7, 2020.
3 Connolly JJ et al. J Clin Transl Hepatol. 2018 Sep 28;6(3):264-275. 5. Liu Z et al. Diabetes. 2016 Dec;65(12):3598-3609.5. 6. Sanz S et al. Gut. 2005; 54:134-141. 7. Stefano JT et al. World J Gastroenterol. 2006 Jun 28; 12(24):3821–3828. 8. Ditchel LE et al. Clin Transl Gastroentrol. 2017; 8:e217